How to Make Enough Vaccine for the World in One Year
By Zoltán Kis and Zain Rizvi
Last September, a biotechnology company bought a manufacturing plant in a small German city.[1] The 300 staff at the facility had never worked with the new vaccine technology used by the company.[2] But, in less than six months, the team switched from making cancer medicines to pumping out vaccines.[3] Today, BioNTech’s Marburg facility is producing millions of mRNA vaccine doses a week.
What would it take to produce enough coronavirus vaccine for the world in one year?
In this report, we provide one path forward. Using computational process modelling, we show how the global community could set up regional hubs capable of producing eight billion mRNA vaccine doses by May 2022.[4] This would be enough to cover 80% percent of the population—what some experts believe is necessary for achieving herd immunity—in low- and middle-income countries.[5] Critically, given the adaptability of mRNA technology, it also would set up the infrastructure required to quickly address variants and future public health threats. This entire analysis is presented for the National Institutes of Health-Moderna Covid-19 vaccine (mRNA-1273), the BioNTech-Pfizer Covid-19 vaccine (BNT162b2), and the CureVac vaccine (CVnCoV).[6]
In the case of the NIH-Moderna vaccine, for example, we estimate that producing eight billion doses in one year would cost $23 billion. 842.1 kg of mRNA would be needed. This would require 4,620 employees working at 55 production lines, which could be set up in 14 facilities. The capital cost for retrofitting facilities would be $3.2 billion, while the operating cost for the drug substance production campaign would be $17.5 billion. Fill and finish would cost $2 billion.[7]
Table 1: Summary of key resource estimates for producing 8 billion doses of mRNA vaccine.
| Resource
|
mRNA-1273
(8 billion 100 μg doses)* |
BNT162b2
(8 billion 30 μg doses)* |
CVnCoV
(8 billion 12 μg doses)* |
| Facilities | 14 | 5 | 2 |
| Production lines** | 55 | 17 | 7 |
| mRNA [kg] | 842.1 | 252.6 | 101.1 |
| Production batches | 10,175 | 3,145 | 1,295 |
| Personnel | 4,620 | 1,386 | 554 |
| Drug substance capital cost | 3.19 billion | 985 million | 405.6 million |
| Drug substance operating cost¥ | 17.48 billion | 5.40 billion | 2.22 billion |
| Fill and finish operating cost¥ | 2.16 billion | 3.04 billion | 2.16 billion |
| Total cost‡ | $22.83 billion | $9.43 billion | $4.38 billion |
* The values of the production requirements were calculated based on producing 8 billion vaccine doses within 6 months, as additional six months are required beforehand to construct, equip, validate, and start up production [8]. ** Drug substance production lines at the 30L bioreactor working volume scale. ¥ This operating cost is calculated for the six month period required to produce 8 billion doses and it includes the annualized facility-dependent capital costs. ‡ We assumed there is sufficient drug product capacity and new facilities did not need to be constructed.
Raw materials are the major cost component for mRNA vaccines. These materials currently cost more than materials for other kinds of vaccines. The significant difference in resource estimates between the three vaccines reflect differences in mRNA dose. mRNA-1273, which uses a 100 μg dose, is the most resource intensive, followed by BNT162b2 (30 μg) and CVnCoV (12 μg). Raw material costs could come down with greater production volume and additional suppliers.
We proceed in three parts. First, we describe the production process for mRNA vaccines, noting in particular the significant advantages of cell-free production. Crucially, mRNA production facilities can be smaller, cheaper, and faster to establish. Many more manufacturing facilities can also be retrofitted to produce mRNA vaccines, compared to other kinds of vaccines, given both the small scale and “closed” nature of the process. Second, we discuss the practical steps required to setup mRNA facilities in six months, including technology transfer and capacity building. Finally, we describe the raw materials, consumables, personnel, and facilities required to produce eight billion drug substance doses within an additional six months and costs, vial and filling line requirements for drug product manufacturing. Together, we describe how the world could produce eight billion doses in one year.
Our analysis is limited by a lack of transparency. None of the companies fully disclose their production process. This model reflects our understanding of the optimal process for mRNA production, based in part on publicly available information, and is intended to convey the magnitude of resources required. A forthcoming Public Citizen analysis will describe the legal authorities the U.S. government could draw on to share technology, remove intellectual property barriers, and address raw material production bottlenecks.
The COVID-19 pandemic has highlighted the urgent need for distributed manufacturing. mRNA technology holds particular promise. Indeed, according to the World Health Organization, 19 manufacturers from more than a dozen countries in Africa, Asia, and Latin America have expressed interest in ramping up mRNA vaccine production.[9] This report presents a technical roadmap on how to end this pandemic—and potentially all others.
2.Description of the mRNA production process
There are two main phases in vaccine production: 1) production of the drug substance (aka. active ingredient production, bulk production or primary manufacturing) and 2) drug product manufacturing (aka. fill and finish, or secondary manufacturing). These two production processes usually happen at different locations and they require different equipment, facilities, quality control methodologies, and expertise. Both the drug substance and drug product manufacturing processes follow strict regulatory guidelines and must comply with current Good Manufacturing Practices (cGMP) in order to ensure product safety, efficacy, and product quality.[10] The steps required for producing mRNA vaccines are shown in Figure 1 below.[11]
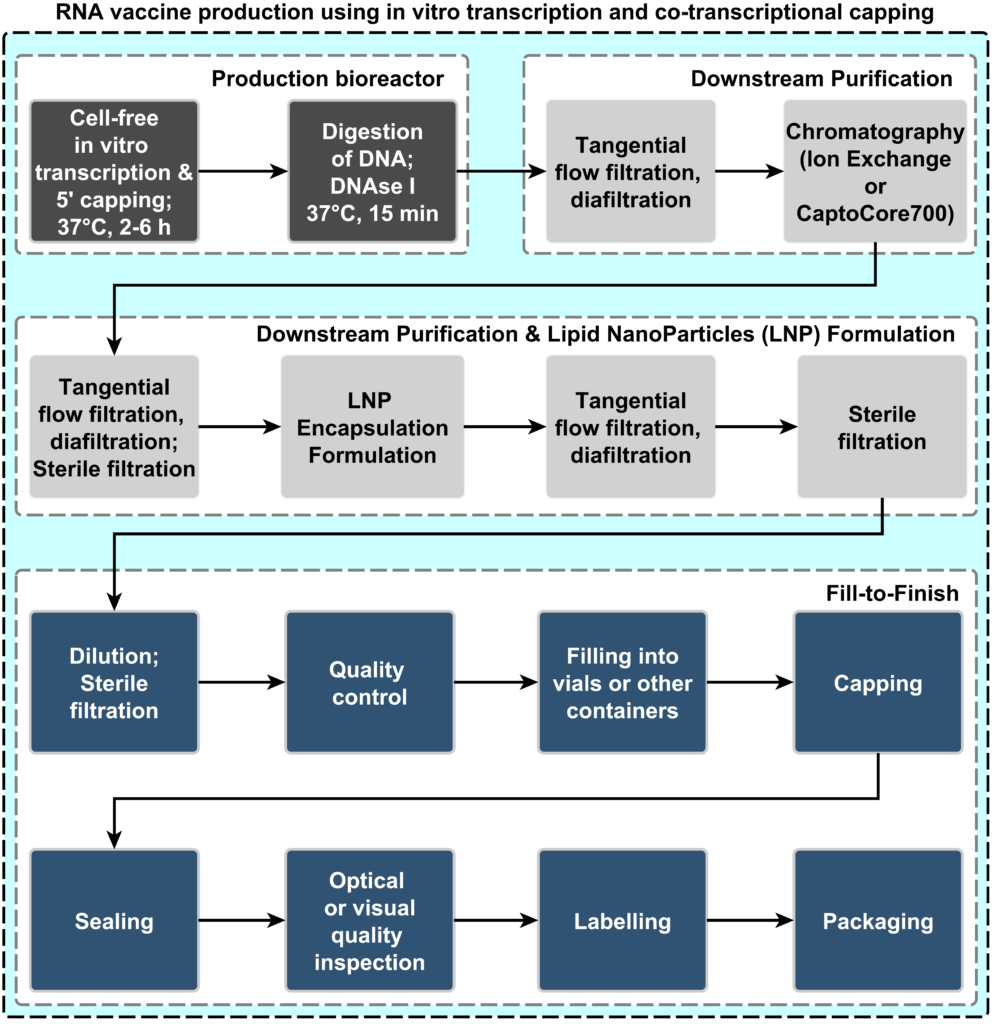
Figure 1: Process flow diagram for mRNA drug substance production (aka. active ingredient production, bulk production, or primary manufacturing) and drug product manufacturing (aka. fill and finish or secondary manufacturing).
Critically, mRNA vaccines offer several advantages over other vaccine platform technologies. Conventional cell-based expression technologies require growing cells in large bioreactors (e.g., 2000L), which depends on a careful optimization of biological conditions. Cells introduce some variability. While mRNA production uses a novel process, it is cell-free and more akin to biochemical synthesis. There are at least seven distinct advantages:
- The process is simpler. mRNA vaccine production requires fewer steps compared to cell-based vaccine production, especially in the upstream processing section. The mRNA vaccine production reaction mix also uses fewer and more well-defined components compared to cell-based vaccine production as the mRNA vaccine production reaction mix does not contain nutrients for cells, cell debris, proteins, chromosomal DNA, lipids and complex sugars that cells release.[12] This simplicity of the reaction mix in turn reduces the complexities in the downstream purification process.[13]
- The process is more robust. Once the mRNA vaccine production process is established, there is also less potential for biological variability. Living cells perform complex, interconnected functions. They can sometimes behave unpredictably, leading to unexpected results such as reduction in production yields.[14] mRNA vaccine production can largely bypass this complexity.
- Dose production is faster. The production of the vaccine active ingredient, the mRNA molecule, can be completed in two to six hours. The entire production batch for mRNA vaccines, including the enzymatic synthesis of the mRNA, purification, and formulation, can be completed within days, excluding the time required for quality control testing.[15] For example, BioNTech produces batches at three to seven day intervals, with four to five weeks spent for quality control.[16] In contrast, completing batches for cell-based vaccines can take several months due to the time required to grow the cells to the specified volumes and quantities.[17] The productivity of the mRNA vaccine production, expressed in doses per L of bioreactor working volumes per day, is two to four orders of magnitude higher compared to most cell-based vaccine production processes.[18]
- The facilities and equipment can be an order of magnitude smaller. mRNA vaccine production can take place in much smaller bioreactors (e.g., 30L to 50L) than those generally employed in cell-based vaccine production (e.g., 2000L).[19] While mRNA vaccine production can require high volume buffer solutions, the smaller scale of the mRNA vaccine production process means it can be implemented in smaller facilities. Multiple mRNA vaccine production processes, for example, can be placed into a conventional cell-based vaccine production facility.
- The capital costs are lower. Given the small-scale nature, setting up the production process and covering facility related expenses is cheaper for mRNA vaccines.
- The process can be implemented in more existing facilities. The number of existing facilities that can accommodate mRNA vaccine production is higher than the number of facilities that can be used generally for setting up new cell-based vaccine production processes. In addition to its small-scale nature, mRNA vaccine production can be hermetically sealed from the environment.[20] This can allow production in lower grade clean rooms.[21] mRNA vaccine production processes can in principle be set up in clean rooms in existing facilities that are used to produce other vaccines, monoclonal antibodies, insulin, veterinary vaccines, and other biologics and injectables.
- The process can be quickly repurposed for new variants, or even new viral threats. mRNA vaccines involve rapid development and production timelines because the production platform is agnostic to the disease target. Different RNA sequences translating into different vaccine or candidate vaccine protein antigen can be produced using the same process.[22] The only component in this production process that needs to be changed is the template DNA based on which the RNA is enzymatically synthesized. The rest of the materials, equipment, consumables, unit operations, formulation components, fill and finish processes as well as quality control and quality assurance methods remain unchanged when switching to the production of a new RNA sequence encoding for a new vaccine antigen. This flexibility can help ensure long-term sustainability.
Potential challenges with scaling up mRNA vaccines include the limited pool of production and quality experts with experience using the new technology, particularly compared to other kinds of vaccines, and challenges in quickly sourcing raw materials (e.g., the cationic lipid) at the quantities required. Nonetheless, the manufacturing advantages of the mRNA vaccine platform have been demonstrated in the COVID-19 response. The mRNA manufacturers are among the few manufacturers that are currently increasing their minimum projections for vaccine dose production by the end of 2021.[23] Moderna partnered with Lonza, an organization with experience in making biologics at commercial scale but limited experience in vaccine production. BioNTech converted a cancer biologics manufacturing facility into a vaccine manufacturing facility and helped train staff in six months.
2.1. Upstream: the in vitro transcription reaction
mRNA vaccines represent an innovation in the drug substance production process. As shown in Figure 1, the mRNA drug substance (DS) production process starts with carrying out the biochemical reaction whereby the mRNA is synthesized.
This in vitro transcription reaction for mRNA vaccine production starts with adding all the reaction components to the bioreactor. These reaction components include:
- Nucleotides (adenosine-5′-triphosphate (ATP), 1-methylpseudouridine-5′-triphosphate (mod-UTP), cytidine-5′-triphosphate (CTP), guanosine-5′-triphosphate (GTP));
- a linear template DNA (can be mass-produced in Escherichia coli, and linearized using restriction enzymes);
- T7 RNA Polymerase enzyme (produced in Escherichia coli) and RNase enzyme inhibitors;
- 5’ cap analogue (e.g. CleanCap AG); and
- spermidine, dithiothreitol (DTT), magnesium chloride (MgCl2), optionally the pyrophosphatase enzyme (in order to break down the pyrophosphate byproduct and consequently to maintain the Mg cofactor concentrations), nuclease-free purified water, and buffers to maintain the pH (e.g. Tris-HCl).
In this reaction, which takes about two to six hours to complete, the T7 RNA Polymerase enzyme links together the four RNA building blocks (ATP, mod-UTP, CTP, GTP) in the sequence provided by the linear template DNA (i.e., the Spike protein of the SARS-CoV-2 virus). The linear chain of nucleotides formed in this reaction constitutes the mRNA of the vaccine. A polyadenylate tail (poly(A) tail) is also encoded on the DNA and added to the mRNA during the in vitro transcription reaction.
The 5’ cap structure (e.g. CleanCap AG), at the beginning of the mRNA, can be incorporated during the in vitro synthesis of the mRNA in an approach called co-transcriptional capping. (While capping can in principle also be done after synthesis, in this report, we modeled co-transcriptional capping.) The 5’ cap is essential for enabling the mRNA to work in the cells of the human body, without being detected as an RNA of foreign (e.g. viral) origin.
The 1-methylpseudouridine-5′-triphosphate (mod-UTP) is used instead of the wild-type, unmodified uridine-5′-triphosphate (UTP) in order to reduce the immunogenicity and increase mRNA translational capacity and biological stability, which is advantageous for mRNA vaccinology.[24] The Moderna and Pfizer vaccines use mod-UTP but the CureVac candidate does not.
Following the synthesis of the 5’ capped mRNA which contains mod-UTP, the linear template DNA is degraded by adding an enzyme which degrades DNA to the bioreactor. For this, the Deoxyribonuclease I (DNase I) enzyme can be used. The activity of DNase I is highest in the presence of Calcium ions, thus calcium chloride (CaCl2) also is added to the bioreactor together with the DNase I enzyme.
2.2. Downstream purification and formulation
Following the digestion of the template DNA, the next task is to purify the product of interest, namely the full length 5’ capped mRNA, out of the aqueous solution containing the above described components and RNA fragments that were obtained by unsuccessful in vitro transcription or as a result of degradation. The downstream separation and purification of the mRNA can be achieved based on differences in size, electrical charge, hydrophobicity, binding affinity to specific ligands, and other properties. Following the digestion of the template DNA, the largest component by molecular mass is the mRNA product at a molecular mass of 2.5 MDa, followed by the enzymes which are one order of magnitude smaller in molecular mass (e.g. the T7 RNA polymerase is ≈0.1 MDa, or ≈100 kDa, whereas DNase I is ≈0.03 MDa, or ≈30 kDa). Therefore, size-based separation, such as filtration, can be used.
The exact purification setup that mRNA Covid-19 vaccine production companies use is not disclosed publicly. Possible purification unit operations include: tangential flow filtration (TFF), ion exchange chromatography, core bead chromatography (e.g. Capto Core 700), oligo dT affinity chromatography, size exclusion chromatography, and hydroxyapatite chromatography.[25] Out of these, TFF, ion exchange chromatography, core bead chromatography (e.g. Capto Core 700) and oligo dT affinity chromatography are likely used.[26]
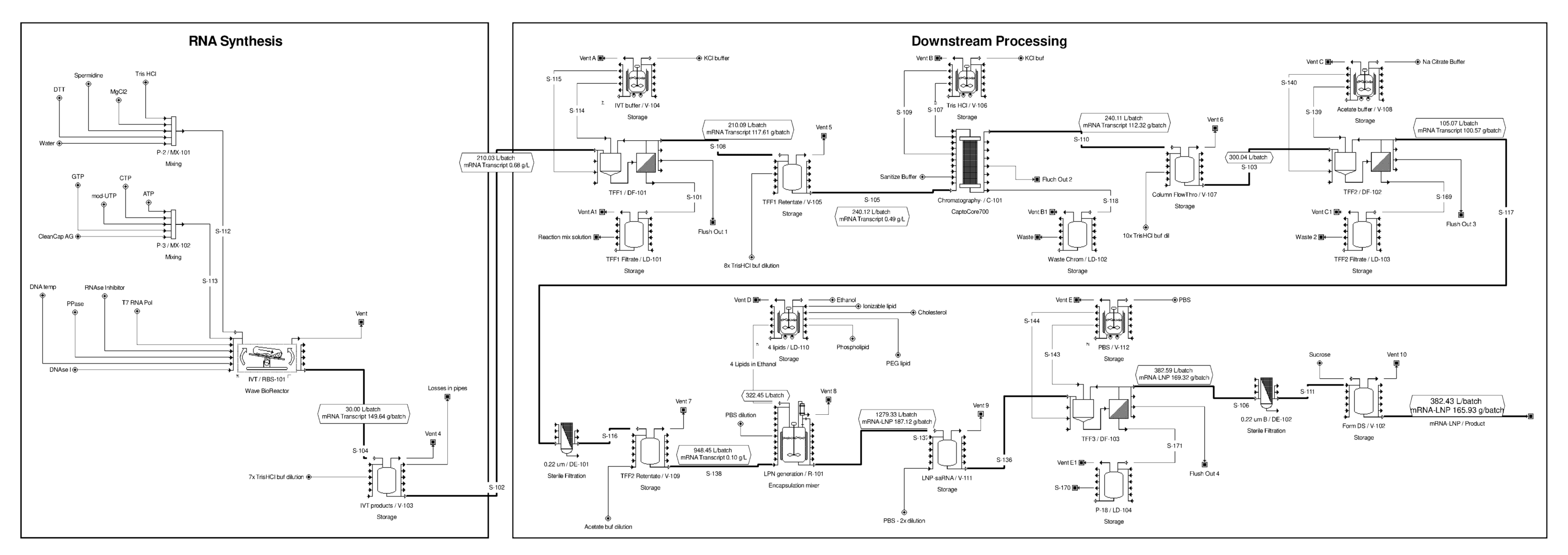
A potential version of this downstream purification process is shown above in Figure 1. Based on this, a detailed process flow model was built in the SuperPro Designer bioprocess simulation tool (Figure 2).[27] In this version of the process, the downstream purification starts with a dilution step followed by the TFF unit operation where the mRNA molecule is retained by the filter and the other, smaller components of the reaction mix flow through the TFF filter membrane. Additional buffer solution (e.g. potassium chloride (KCl) buffer) is added to the solution retained by the filter to wash away the impurities through the filter membrane more effectively. The amount of added buffer solution is 10-fold the volume of the solution entering the TFF diafiltration step (i.e., 10x diafiltration volume). For the TFF steps presented here, ultrafiltration membranes with a molecular weight cut-off of 500 kDa or 300 kDa would be suitable.
In addition to washing away the impurities, in this TFF step the mRNA is placed into a solution which is suitable for the next purification step. The solution retained by the TFF membrane is then flown into the Capto Core 700 chromatography unit operation which is a core bead type chromatography whereby molecules can be separated based on both size and binding to hydrophobic and positively charged octylamine ligands.[28] The advantage of using this chromatography approach is that lower amounts of chromatography medium/resin can be used to purify the mRNA product more efficiently.
After the mRNA flows through the CaptoCore 700 chromatography column, it enters a second TFF unit operation whereby it is concentrated and then further washed using a 10x diafiltration volume. The impurities are further washed away, and the buffer is again replaced, this time with a sodium citrate buffer. Next the mRNA in citrate buffer is sterile filtered. It then enters the formulation step, where the mRNA is enclosed into spheres of lipids called lipid nanoparticles (LNPs).
This LNP formulation step requires the mixing of a liquid stream containing four lipid components in ethanol with another liquid stream containing the mRNA in the water-based sodium citrate buffer. The four lipids are: ionizable lipid (this is the most novel component), the phospholipid, cholesterol, and a polyethylene glycol (PEG)-lipid conjugate. The LNP formulation step can be carried out using microfluidics mixers,[29] jet-impingement mixers (aka. T-junction mixers), [30] or potentially in pressurized tanks.[31] For fast and large-scale production mixing in pressurized tanks is the most preferred option, whereas microfluidics mixing is the least preferred option.[32] In the model presented here, 14 hours were assumed for the LNP encapsulation reaction in macro-scale process similar to the BioNTech/Pfizer approach.[33] The entire formulation unit operation with setting up the equipment, transferring in solutions, performing the 14-hour long formulation reaction and cleaning was modelled to take 19.24 hours in total.
The duration of this LNP formulation unit operation is not publicly available and can vary among the mRNA vaccine manufacturing companies. This step is considered to be the bottleneck in the mRNA drug substance production process, and consequently the duration of this step can impact production performance of the process in terms of costs and annual production amounts.
The time required to complete one production batch was modelled at 58.78 hours. By optimizing the scheduling, batches can be completed at 22.25-hour intervals (i.e., if the production of a batch starts in the upstream section, while the previous batch is being completed in the downstream and formulation sections of the production process). Following the LNP encapsulation step, the LNP-mRNA particles are first concentrated and then washed in a diafiltration step. The ethanol and other impurities are washed away and the buffer is replaced with the final formulation buffer, for example by using 10x diafiltration volumes. After this, the LNP-mRNA containing solution is sterile-filtered and filled into plastic bags (e.g. 10 L plastic bags) which are then sent to the fill and finish facilities.
The approach described here represents one possible way of purifying the synthesized mRNA and the mRNA-LNP complexes. Companies such as Moderna/Lonza, BioNTech/Pfizer, and CureVac might use different purification approaches; however, the goal remains the same: to separate the mRNA product of interest away from other impurities in the process in order to ensure product quality, safety, and efficacy, using cGMP-compliant processes.
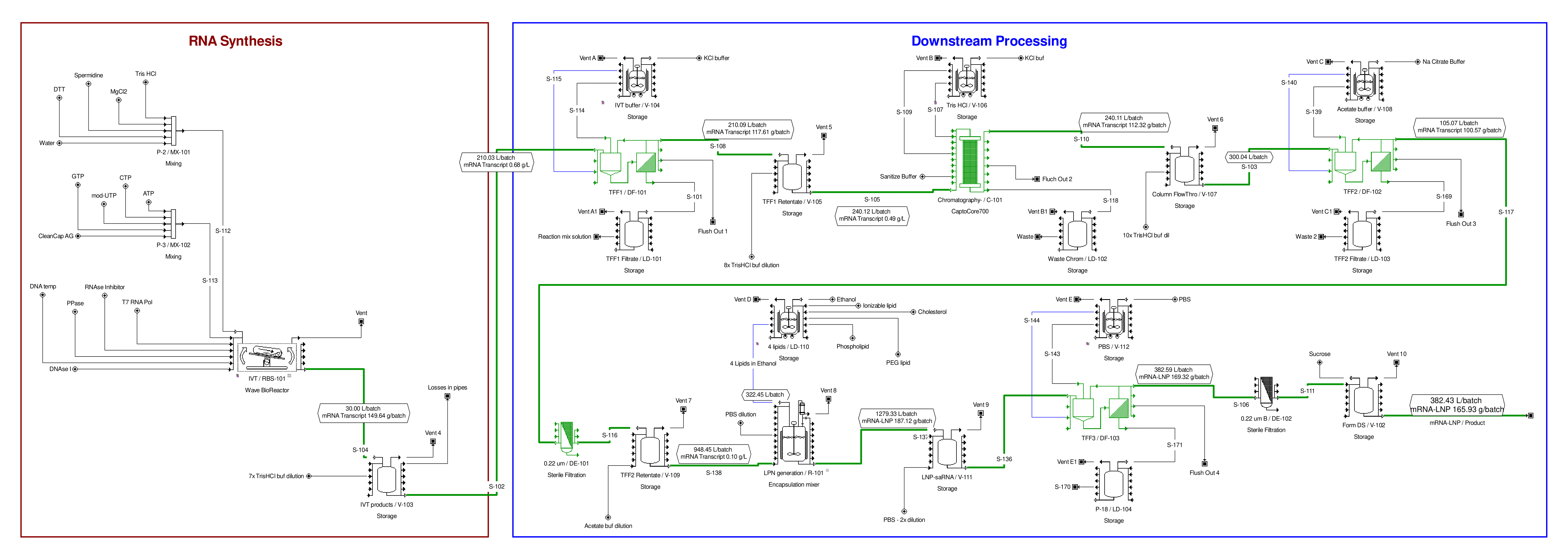
Figure 2. mRNA vaccine drug substance production process flow diagram obtained from SuperPro Designer. High-resolution diagram available here.
The production process consists of the following three key parts: 1) the in vitro transcription step, whereby the mRNA is capped co-transcriptionally using 5’ cap analogues (e.g. CleanCap AG); 2) downstream purification step obtained by a series of tangential flow filtration steps and a Capto Core 700 chromatography step; 3) LNP formulation step followed by another tangential flow filtration step. The tangential flow filtration steps can perform both concentration and diafiltration of the mRNA product.
2.3. Fill and finish and quality control
At the fill and finish facilities the LNP-mRNA particles can be optionally further purified and sterile filtered. At this point quality control testing of the drug substance and of the sterilized vials can also take place. Next, if required, the LNP-mRNA solution can be diluted to the concentration in which the solution is placed into glass vials. NIH-Moderna (mRNA-1273) vaccine is filled into 10-dose glass vials whereas the BioNTech-Pfizer (BNT162b2) vaccine is filled into six-dose glass vials. For this, the sterile and clean glass vials are moved along a conveyor and a liquid dispensing needle fills the formulated LNP-mRNA into the glass vials from the top in a sterile environment. Next, the vials are capped, sealed, and inspected for visual defects (this can be achieved using cameras and automated image processing). The vials without defects are then labelled and packaged in several (e.g. two additional) layers of packaging.
Since both the drug substance and drug product manufacturing processes are compliant with cGMP, the raw materials, the in-process materials, and finished products are strictly monitored for their quality. The quality control and batch release can take several weeks to complete (e.g., four to five).[34] To ensure uniform delays between production times and batch release, the production runs can be staggered and the quality control for subsequent production runs can be carried out in parallel.
3. Roadmap for implementation
This report lays out a path for significantly increasing distributed mRNA vaccine production in one year. We previously described the mRNA production process and its advantages. Below, we describe what it would take to establish new production capacity within six months.
We draw, in part, on a recent example. In September 2020, BioNTech acquired a GMP certified biopharmaceutical manufacturing plant (with no built-in mRNA vaccine production capacity) from Novartis in Marburg, Germany and adapted it for mRNA vaccine production, starting commercial scale production in February 2021.[35]
The roadmap for setting up distributed mRNA vaccine manufacturing in regional hubs, as described above, can be divided into four phases: 1) Early planning; 2) Advanced planning, facility improvement, and technology transfer initiation; 3) Technology transfer and process set-up; and 4) Facility and process validation & start up.
3.1. Early planning phase (1-2 months)
In the first phase, in order to streamline the expansion of the mRNA vaccine manufacturing capacity in the shortest possible timeframe, a detailed action plan should be developed by involving all the relevant parties and experts from the beginning, as soon as funding is committed to the project. At this planning phase, an initial risk assessment should also be carried out and a business plan can be developed. In parallel, experts should be recruited, including experts for manufacturing, logistics, technology transfer, and quality control. In order to build up the expertise, the personnel should be trained to fill all the expertise gaps, and to familiarize the personnel with the specifics of the mRNA vaccine production process and quality testing procedures. Training of the personnel can be done in a centralized manner, for example at a governmental or international non-governmental organization (e.g., World Health Organization).
Regulatory authorities or regulatory experts should be involved and consulted at this early stage in order to ensure that the project follows the relevant regulatory guidelines from the beginning. This can help avoid potential delays down the line.
Candidate locations/sites, where the mRNA vaccine platform technology will be transferred into, should be assessed. Setting up mRNA vaccine production processes based on single-use equipment in existing facilities could help to shorten the timelines required for building up capacity. The facilities where the mRNA vaccine production process will be transferred should meet the requirements of the process in terms of sterility/clean room grade, regulatory requirements, floor space at the required sterility/clean room grades, utilities and WFI production, and safety requirements (e.g. for working with flammable and explosive ethanol solutions, which is required for the LNP formulation step).
In parallel, technology transfer teams should be established in order to cover both the sending and receiving site. The team should have expertise in project management (to lead the technology transfer team), business aspects (finance, legal, sales, strategy), production engineering and process validation (assuring that the production process produces the required product), analytical (for raw materials, in-process controls, product quality and product stability), engineering (facility construction, equipment installation and qualification), manufacturing (plant operation), regulatory, quality (quality management systems and compliance), sourcing and supply chains (material & consumable sourcing, production planning, logistics). Having the know-how, experience and skills of the original technology developers available can help avoid errors and duplication of previous work and activities (e.g. to avoid reproduction and reverse engineering of various processes and aspects).
The materials, consumables, manufacturing equipment and analytical equipment should be ordered as soon as possible.
3.2. Advanced planning, facility improvement and technology transfer initiation phase (1-2 months)
The second phase would involve continuing to train experts, evaluating the receiving facilities/sites, updating the risk assessment, working with regulatory authorities/experts, and coordinating with key suppliers.
At this stage a detailed understanding of the production process to be transferred in should be available. The relevant analytical and quality control assays should be in place for raw materials, in-process controls, product quality, product stability. Quantifiable acceptance criteria should be set for the vaccine product and technology transfer process.
Once the assessment of the facilities and locations has been completed (this task was started in phase 1), the facilities/sites selected to receive the technology should be adapted (if needed) to meet the requirements of the process and to meet regulatory requirements. For this, the necessary facility/site upgrades/improvements should be first planned and then implemented. This work should ensure that the receiving facility will have the required utilities, including electricity supply (also back-up generators) and WFI production capacity, waste disposal, and that it will meet the safety requirements for working with ethanol, which is flammable. If extensive works are required, such as yard and site improvements (e.g. excavation, site grading, roads, fences, etc.) for building facilities using pre-fabricated modules, then some of these works will take longer to complete.[36]
3.3. Technology transfer and process set up phase (1-2 months)
At the beginning of the third phase, the repurposed facilities should be evaluated and a new site-specific risk assessment for the project should be carried out. The production equipment, consumables, materials, and analytical equipment should be received from the suppliers. Once received and tested, this equipment should be installed. Once the process is configured it should be tested by running so-called engineering batches. Since the mRNA vaccine production platform is based on a new technology, which is used at the production scale for the first time during the pandemic, extra time and funding should be allowed for troubleshooting and additional tasks that might be required.
3.4. Facility and process validation, start-up phase (1-2 months)
Once the process has been set up in a facility, following the relevant regulatory guidelines, the fourth, process validation phase could start.[37] For prospective and concurrent process validation, the production of three consecutive successful production batches is generally required, although there may be situations where additional process runs are required to prove consistency.[38] The overall aim is to prove that the process can produce products consistently at high quality. Once the process has been validated and approved by the regulatory authorities, and once the cGMP certification is received, the production of mRNA vaccine can begin.
The entire project of establishing new manufacturing facilities as outlined in this section can be completed in 5-6 months when the mRNA vaccine production process is placed into existing facilities[39] and in 6-8 months if new facilities are constructed using pre-fabricated modules.[40]
4. Resources required for producing eight billion doses
In this part, we model the resources required to complete a production campaign for eight billion doses in six months using SuperPro Designer Version 11, Build 2 from Intelligen, Inc, assuming batch operation mode.[41] The SuperPro Designer bioprocess simulation tool is used by industry experts to calculate material and energy balances for each unit operation in the production process.[42] SuperPro Designer determines the size of the equipment, calculates labor requirements, schedules the operations and procedures, and performs economic calculations both for capital expenses (CapEx) and operating expenses (OpEx). SuperPro Designer is linked to databases of chemicals, consumables, equipment, and other resources.
The results presented in this report are based on previously published work by one of the authors.[43] The biomanufacturing models presented have been updated to include the latest developments in this rapidly evolving field.
Table 2: List of mRNA vaccines considered in this study and the amount of mRNA required for 8 billion doses.
| Developer | Vaccine name | mRNA per dose [µg/Dose] | Doses per person | mRNA required for 8 billion doses [kg]* | Ref. |
| NIH-Moderna Inc. | mRNA-1273 | 100 | 2 | 842.1 | [44] |
| BioNTech SE; Pfizer Inc. | BNT162b2 | 30 | 2 | 252.6 | [45] |
| CureVac N.V. | CVnCoV | 12 | 2 | 101.1 | [46] |
* The amount of mRNA required for 8 billion doses was calculated by multiplying the mRNA amount per dose by 8 billion and by accounting for the 5% losses assumed in the fill and finish.
Producing eight billion doses of the NIH-Moderna Covid-19 vaccine (mRNA-1273), which contains 100 µg of mRNA per vaccine dose requires in total 842.1 kg of mRNA to be produced. Eight billion doses of the BioNTech-Pfizer Covid-19 vaccine (BNT162b2) require the production of 252.6 kg of mRNA, whereas 101.1 kg of mRNA would yield eight billion doses of the CureVac (CVnCoV) mRNA vaccine, as shown above in Table 2. To calculate the amount of mRNA, a 5% overproduction was considered to account for the losses that were assumed to occur in the subsequent fill and finish processes.
4.1. Facility-related requirements for drug substance production
The facility-related operating costs amount to approximately 1% of the total production operating costs. This is different from conventional, cell-based vaccine production technologies whereby the facility-related costs tend to represent the major operating cost component.
There are two main ways of rapidly increasing mRNA vaccine production capacity: 1) setting up vaccine production processes in existing cGMP compliant facilities which are used for the production of other vaccines, monoclonal antibodies, insulin, veterinary vaccines, and other biologics and injectables, based on the approach taken by BioNTech in Marburg, Germany[47] or 2) using pre-fabricated modules to build facilities that can be quickly assembled on site and then equipped, based on the approach taken by Pfizer in Kalamazoo, Michigan.[48] Pfizer, for example, used, modular units that were pre-fabricated in Texas and then shipped to Kalamazoo where they were assembled.[49] In both cases, the mRNA vaccine production process was established largely based on single-use equipment.
The exact number of production lines that could fit into existing facilities needs to be determined on a case-by-case basis by considering the details and requirements of the process and footprint of the facility, the sterility/clean room grade of the rooms, and the utilities, among other things. In this report, we assumed that placing three or four mRNA vaccine production processes at the 30L bioreactor working volume scale into an existing facility was feasible given that the mRNA production process is at a smaller scale compared to conventional vaccine production processes for which the bioreactor is commonly at the 2000L scale.
To produce eight billion doses of the NIH-Moderna Covid-19 mRNA vaccine within six months, 55 production lines would be required, as shown in Table 3 below. These 55 production lines can be placed into 14 facilities and would complete a total of 10,175 batches in six months. Out of the 14 facilities, 13 facilities would contain four production lines per facility and the remaining one facility would contain three production lines per facility. The total capital cost required to construct, equip, validate, and start up the 14 facilities, with three or four production lines per facility, for producing eight billion mRNA-1273 vaccine doses is estimated at $3.19 billion.
To produce eight billion doses of the BioNTech Covid-19 mRNA vaccine within six months, 17 production lines and 3,145 batches would be required, as shown in Table 3 below. These 17 production lines can be placed into five facilities, three of these facilities would have three production lines per facility and the remaining two facilities would contain four production lines per facility. The total capital cost required to construct, equip, validate, and start up the five facilities, with three or four production lines per facility, for producing eight billion BNT162b2 vaccine doses is estimated at $985 million.
To produce eight billion doses of the CureVac Covid-19 mRNA vaccine within six months, seven production lines and 1,295 batches would be required, as shown in Table 3 below. These seven production lines can be placed into two facilities; one of these facilities would have three production lines and the other facility would contain four production lines. The total capital cost required to construct, equip, validate, and start up the two facilities with three or four production lines per facility for producing eight billion CVnCoV vaccine doses is estimated at $405.6 million.
Placing more than one production line into the same facility can reduce the overall facility-related costs per amount of produced product, because some facility-related resources can be shared between the production lines within the same facility, compared to placing one production line into a dedicated facility. Therefore, having 3 or 4 production lines versus 1 production line per facility would result in a slight decrease of facility-related costs, and this has already been accounted for in the current model. The capital costs, annual operating costs, and doses produced per year, expressed per production line are shown below in the Appendix in Table S2.
If the mRNA vaccine production process can be sealed from the environment, and if it can be operated as a closed production system,[50] the above described investment costs can be further reduced. Alternatively, the above mentioned upfront capital investment costs can also be reduced if the production processes will be set up in existing facilities which are adequate for mRNA vaccine production and if the facilities meet the needs of the process (e.g. in terms of utilities and safety).
Table 3: Facility-related requirements for producing 8 billion doses of mRNA vaccine drug substance.
| Name of facility-related resource | Facility-related requirements for producing 8 billion mRNA-1273 doses* | Facility-related requirements for producing 8 billion BNT162b2 doses* | Facility-related requirements for producing 8 billion CVnCoV doses* |
| Number of production lines‡ | 55 | 17 | 7 |
| Number of facilities | 14 | 5 | 2 |
| Production lines per facility | 4 production lines for 13 facilities; 3 production lines in 1 facility | 4 production lines for 2 facilities; 3 production lines for 3 facilities | 4 production lines for 1 facilities; 3 production lines for 1 facility |
| Total number of batches required | 10,175 | 3,145 | 1,295 |
| Total operating costs for drug substance ¥ | 17.48 billion | 5.40 billion | 2.22 billion |
| Total capital costs | 3.19 billion | 985 million | 405.6 million |
* The values of the production requirements were calculated based on producing 8 billion vaccine doses within 6 months, as additional 6 months are required beforehand to construct, equip, validate, and start up production [51]. ‡ Production lines at the 30L bioreactor working volume scale. ¥ This operating cost is calculated for the 6 months period required to produce 8 billion doses and it includes the annualized facility-dependent capital costs.
4.2. Materials used in the drug substance production process
Materials represent the major cost component of mRNA vaccine drug substance production, accounting for approximately 73% of the total production costs. Based on process knowledge and input from other experts, we estimate that there are mRNA losses in the production process, including 30% in the downstream purification, 20% in the formulation and subsequent purification steps, and up to 5% in the processes occurring at the fill and finish sites. This translates to a total of approximately 53% losses in the entire production process. Therefore, the raw material amounts used in the production process are higher in order to account for these losses. For example, the quantity of raw materials entering the in vitro transcription bioreactor are nearly double the amount needed to produce the equivalent mRNA amount when not accounting for the losses.
Some of the materials used for mRNA vaccine drug substance are also new, and lack a diversified supply chain, with a limited number of suppliers providing these key materials. We solely focus on these key materials. For example, we did not model plasmid DNA production, because it is better known to industry, has been previously scaled compared to the production of some key mRNA vaccine production ingredients (e.g. CleanCap AG), and can be purchased from a variety of suppliers.
A comprehensive list of materials and their amounts estimated to be used for drug substance production is listed in Table S3 in the Appendix, expressed per production line and for producing eight billion doses of the NIH-Moderna (mRNA-1273), BioNTech-Pfizer (BNT162b2), and CureVac (CVnCoV) vaccines. The estimated costs of these materials is listed in Table S4, expressed per production line and for producing 8 billion doses of mRNA-1273, BNT162b2, and CVnCoV vaccines.
4.2.1. Raw materials used in the in vitro transcription reaction
The key raw materials used in the in vitro transcription reaction that are thought to be in limited supply include the 5’ cap analogue (e.g. CleanCap AG) and modified nucleotides.[52] In addition, due to sudden increased demand suppliers might be struggling to produce sufficient quantities of the T7 RNA polymerase, the linearized template DNA, the DNase I enzyme and the RNAse Inhibitor. However, the production of these components is based on well-established and scalable processes (e.g. fermentation in Escherichia coli). If 5’ capping of the mRNA is carried out enzymatically, the amounts of capping enzymes should be considered instead of the 5’ cap analogues (e.g. CleanCap AG). The amounts of these materials required per production line (at the 30L bioreactor working volume scale), and the material amounts as well as the costs required for producing 8 billion doses is shown below in Table 4.
Table 4: The estimated amounts and costs of key raw materials used in the in vitro transcription reaction for mRNA vaccine production.
| Name of pure component | Amount per year per production line* [g/year] | Key material requirements for producing 8 billion mRNA-1273 doses | Key material requirements for producing 8 billion BNT162b2 doses | Key material requirements for producing 8 billion CVnCoV doses | ||||||
| Amounts
[g / 8 billion doses] |
Costs
[USD / 8 billion doses] |
Amounts
[g / 8 billion doses] |
Costs
[USD / 8 billion doses] |
Amounts
[g / 8 billion doses] |
Costs
[USD / 8 billion doses] |
|||||
| CleanCap AG | 50,972 | 1,471,906 | 5,887,608,320 | 441,572 | 1,766,282,496 | 176,629 | 706,512,998 | |||
| Deoxyribonuclease I (DNase I) | 28 | 809 | 426,336 | 243 | 127,901 | 97 | 51,160 | |||
| RNase enzyme inhibitor | 139 | 4,014 | 3,411,413 | 1,204 | 1,023,424 | 482 | 409,370 | |||
| T7 RNA polymerase | 185 | 5,342 | 728,176,702 | 1,603 | 218,453,011 | 641 | 87,381,204 | |||
| linear template DNA | 555 | 16,027 | 1,762,928,524 | 4,808 | 528,878,557 | 1,923 | 211,551,423 | |||
| 1-methylpseudouridine-5′-triphosphate (mod-UTP) | 21,496 | 620,735 | 2,931,813,779 | 186,220 | 879,544,134 | 0 | 0 | |||
* Production line at 30L bioreactor working volume scale, producing 29162 grams of mRNA per year. For further details see Tables S3 and S4 in the appendix.
4.2.2. Raw materials used for lipid nanoparticle formulation
Each of the three Covid-19 mRNA vaccines presented in this study is formulated in lipid nanoparticles (LNPs).[53] These LNPs are spheres composed of four different lipids with the mRNA enclosed inside.[54] The four types of lipids building up these spheres are: ionizable lipids (i.e., cationic lipids), cholesterol, phospholipids, and polyethylene glycol (PEG) lipid.[55] These classes of lipids are consistent for the 3 mRNA vaccines, as shown in Table 5, however the individual lipids vary among these vaccines, as shown in the footnote below Table 5. It is believed that BioNTech-Pfizer and CureVac use a similar formulation approach, based on the ionizable lipid (ALC-0315, (4-hydroxybutyl)azanediyl)bis(hexane-6,1-diyl)bis(2-hexyldecanoate)).[56] On the other hand, the NIH-Moderna candidate uses a different ionizable lipid ((SM-102, heptadecan-9-yl 8-((2-hydroxyethyl)(6-oxo-6 (undecyloxy)hexyl)amino)octanoate) and has a different lipid composition.
The NIH-Moderna (mRNA-1273) vaccine contains 1.93 mg of lipids in total per vaccine dose.[57] However, the exact amount of the 4 constituent lipids is not disclosed. Here, the amount of the 4 lipids in the mRNA-1273 vaccine were estimated by assuming the same lipid ratios as in the BNT162b2 vaccine. The total amount of lipids required to produce 8 billion doses of the mRNA-1273 was estimated at 20.34 metric tonnes, and over half of this amount consisted of the ionizable lipid.
The BioNTech-Pfizer (BNT162b2) vaccine contains 0.77 mg of lipids per vaccine dose, with known amounts of the four lipid types.[58] The ratio of lipid to mRNA is higher in the BNT162b2 compared to the mRNA-1273 vaccine. In total, it is estimated that 8.13 metric tonnes of lipids would be required to produce 8 billion BNT162b2 doses. The amounts of ionizable lipids, phospholipid, cholesterol, and PEG lipid for 8 billion BNT162b2 were estimated at 4.54, 0.95, 2.11, and 0.53 metric tonnes, respectively.
The amount of lipids contained in a dose of CureVac (CVnCoV) were not available publicly. However, this was estimated based on similarity in lipid composition to the BNT162b2 vaccine and by taking into account the difference in the amount of mRNA per dose of these two vaccines. Thus, the total amount of lipid per dose of CVnCoV was estimated at 0.31 mg. The total amount of lipids required to produce 8 billion CVnCoV doses was estimated at 3.25 metric tonnes. The amounts of ionizable lipids, phospholipid, cholesterol, and PEG lipid for 8 billion CVnCoV were estimated at 1.82, 0.38, 0.84, and 0.21 metric tonnes, respectively.
Due to the lack of detailed information regarding the formulation and purification processes for the different vaccines, it was assumed that 32% of the lipids are lost during the formulation and purification steps for all three vaccines. In reality, the percentage of losses might differ between these three vaccines due to differences in the formulation (e.g. microfluidics vs. macrofluidics) and subsequent purification unit operations.
Table 5: Estimated amounts of lipid components required for producing 8 billion mRNA vaccine doses.
| mRNA Vaccine | Lipid Name | Lipid amount per dose [mg] | Lipid amount for 8 billion doses [metric tonnes] * | Ref |
| NIH-Moderna;
mRNA-1273 |
Ionizable lipid a | 1.09 | 11.48 | [59] |
| Phospholipid b | 0.24 | 2.49 | ||
| Cholesterol | 0.47 | 4.96 | ||
| PEG lipid c | 0.13 | 1.42 | ||
| Total lipids | 1.93 | 20.34 | ||
| BioNTech-Pfizer; BNT162b2 | Ionizable lipid d | 0.43 | 4.54 | [60] |
| Phospholipid e | 0.09 | 0.95 | ||
| Cholesterol | 0.2 | 2.11 | ||
| PEG lipid f | 0.05 | 0.53 | ||
| Total lipids | 0.77 | 8.13 | ||
| CureVac;
CVnCoV |
Ionizable lipid g | 0.17 | 1.82 | [61] |
| Phospholipid h | 0.04 | 0.38 | ||
| Cholesterol | 0.08 | 0.84 | ||
| PEG lipid i | 0.02 | 0.21 | ||
| Total lipids | 0.31 | 3.25 |
a heptadecan-9-yl 8-((2-hydroxyethyl)(6-oxo-6-(undecyloxy)hexyl)amino)octanoate (SM-102); b 1,2-distearoyl-sn-glycero-3-phosphocholine (DSPC); c 1,2-dimyristoylrac-glycero-3-methoxypolyethylene glycol-2000 (PEG2000 DMG); d (4-hydroxybutyl)azanediyl)bis(hexane-6,1-diyl)bis(2-hexyldecanoate) (ALC-0315); e 1,2-distearoyl-sn-glycero-3-phosphocholine; f 2[(polyethylene glycol)-2000]-N,N-ditetradecylacetamide; g probably (4-hydroxybutyl)azanediyl)bis(hexane-6,1-diyl)bis(2-hexyldecanoate) (ALC-0315); h 1,2-distearoyl-sn-glycero-3-phosphocholine (DSPC); i probably 2[(polyethylene glycol)-2000]-N,N-ditetradecylacetamide. * It was assumed that 32% of the lipids are lost in the production process.
It also is worth noting that approximately 17.8k metric tonnes of water for injection (RNAse free purified water) is required per year per production line (Table S3). This water for injection (WFI) requirement should be taken into account when designing the utilities and systems for generating the WFI. The total WFI requirements for producing 8 billion doses is estimated at 514.4k metric tonnes, 154.3k metric tonnes, and 61.7k metric tonnes for the mRNA-1273, BNT162b2, and CVnCoV vaccines respectively.
The total estimated annual purchase price of materials per production line at the 30L bioreactor working volume scale is $456.6 million per year, accounting for approximately 73% of the total production costs. Within this material cost amount, approximately 45% is the CleanCap AG purchase price. In total the materials are estimated to cost $13.2 billion, $4 billion, and $1.6 billion when producing 8 billion doses of the mRNA-1273, BNT162b2, and CVnCoV vaccines respectively.
4.3. Consumables and equipment used in the drug substance production process
The second highest cost component in the mRNA vaccine drug substance production process is the cost of consumables, accounting for approximately 24% of the total production costs. The mRNA vaccine production process can be based on single-use equipment and consumables, which are replaced after every production batch. Single use equipment and consumables include:
- Single-use plastic bioreactor lining bags;
- Single-use plastic storage bags;
- Plastic (e.g. silicone) tubing, single-use aseptic connectors, clamps; and
- Disposable filter membranes, single-use filter assemblies and chromatography columns.
In some cases, filter membranes and certain types of chromatography resins can be re-used, helping reduce costs. However, the re-use of these components has to be validated. The single-use consumables are usually held in place by more permanent structures (e.g., a single-use plastic bioreactor lining bag is placed inside a cylindrical steel support frame which can have glass or plastic windows).
The advantage of using single use equipment is that the process can be assembled substantially faster compared to setting up stainless steel equipment. The upfront capital investment costs for single-use equipment is lower compared to the permanent stainless-steel equipment, but the operating costs could increase in case of single-use equipment compared to stainless steel equipment. In addition, because the single-use based production requires substantially less cleaning, it can reduce labor costs as well as water and cleaning agent requirements, minimize cleaning validation, and increase production speeds.
mRNA vaccine production at the 30L bioreactor working volume scale would require around 150 million worth of consumables per year based on the process-cost modelling results obtained with SuperPro Designer, using the model presented in Figure 2. The cost of consumables required to produce 8 billion doses of the mRNA-1273, BNT162b2, and CVnCoV vaccines is estimated at $4.3 billion, $1.3 billion, and $520 million, respectively.
The amounts of chromatography resins and TFF membranes were also estimated, assuming that 5 g of mRNA can be purified per m2 of TFF membrane, 50 g of mRNA can be purified per L of flow through chromatography medium (e.g. Capto Core 700), 1.8 g of mRNA can be purified per L of oligo dT resin [62], and that 5 g/L of mRNA can be purified per L of multimodal chromatography resin that combines hydrogen bonding and anion exchange chromatography (e.g. Prima S). These values were obtained from the suppliers of the consumables and from discussions with biomanufacturing experts. For TFF it also was taken into account that 3 different TFF unit operations were used in total for mRNA purification and post-formulation purification. The amount of these consumables required to produce 8 billion doses is shown below in Table 6. It is worth noting that some of these consumables (e.g. TFF membranes, oligo dT resins, and Prima S resins) can be reused for multiple batches and this would reduce the required amounts for these consumables. However, re-using these consumables needs to be rigorously tested and validated.
Table 6: The estimated amounts of consumables required for producing 8 billion mRNA vaccine doses.
| Name of key consumables and their unit of measurement for the amount | Key consumable requirements for producing 8 billion mRNA-1273 doses | Key consumable requirements for producing 8 billion BNT162b2 doses | Key consumable requirements for producing 8 billion CVnCoV doses |
| Tangential flow filtration membrane [m2] | 505,262 | 151,579 | 60,631 |
| Flow through chromatography medium, e.g. Capto Core 700 [L] * | 16,842 | 5,053 | 2,021 |
| Oligo dT chromatography resin [L] * | 467,836 | 140,351 | 56,140 |
| Multimodal chromatography, hydrogen bonding and anion exchange chromatography, e.g. Prima S [L] * | 168,421 | 50,526 | 20,210 |
* Out of these three types of chromatography resins one or potentially two can be enough to purify the mRNA drug substance in combination with tangential flow filtration.
4.4. Labor required in the drug substance production process
The labor cost in the mRNA vaccine production process represents less than 3% of the total production costs. Based on the modelling results obtained from SuperPro Designer, using the model presented in Figure 2, the total annual operator labor requirement is 113,186 hours per production line at the 30L bioreactor working volume scale. The basic labor rate for the production process operators was assumed at $35 per hour. After accounting for the benefits, supplies, supervision, and administration, the total labor cost was estimated at $80.5 per hour.
The cost of labor can vary among the different geographical locations, countries, and continents but the cost of labor is not expected to have a substantial impact on the overall production costs. In the SuperPro Designer modelling tool, it also was assumed that 60% of the labor hours are used directly for producing the product and the remaining 40% is used for other activities.[63]
In order to calculate the number of personnel required to operate the production process, the total annual 113,186 labor hours required per production line were divided by the average hours that one person works per year (2,760 hours). The value was obtained by multiplying a high 345 working days per year per person (due to the high pandemic-induced global vaccine demand) by eight working hours per day per person. This resulted in requiring around 41 persons to operate each production line. However, Lonza reported that it required over 70 employees per production line.[64] The difference between the personnel requirements from Lonza and the values obtained by the model may be due to the labor required to carry out the quality control testing, warehousing for incoming and outgoing materials, logistics, and administration. To avoid underestimating the labor requirements, the values shown below in Table 7 were updated with the assumption that 80 employees are required per production line.
To produce 8 billion doses of the mRNA-1273, BNT162b2, and CVnCoV vaccines within six months, an estimated 6.38 million labor hours, 1.91 million labor hours, and 765.12 thousand labor hours, respectively, will be required. This translates to 4,620 persons, 1386 persons, and 554 persons required respectively, as shown below in Table 7.
Table 7: Labor required for producing 8 billion mRNA vaccine doses.
| Name | mRNA-1273* | BNT162b2* | CVnCoV* |
| Total number of hours for producing 8 billion doses | 6,375,988 | 1,912,796 | 765,119 |
| Total number of persons for producing 8 billion doses* | 4,620 | 1,386 | 554 |
| Total labor costs for producing 8 billion doses* | 514 million | 154 million | 62 million |
* The values of the production requirements were calculated based on producing 8 billion vaccine doses within 6 months, as an additional 6 months are required beforehand to construct, equip, validate and start-up production.[65] The labor required to construct, equip, validate and start-up the production process is not included in this analysis.
4.5. Other resource requirements for drug substance production
The remaining resource requirements include: 1) laboratory, quality control, and quality assurance; 2) utilities; 3) sales resources; 4) waste treatment & disposal, failed product disposal; 5) transportation and miscellaneous. In total, these were estimated to account for less than 1% of the total production costs. Out of these, laboratory, quality control and quality assurance costs are the highest cost component and were assumed to account for 50% of the total labor costs. In addition, assessing royalties and licensing fees for some components, including the cationic lipids used in the LNP formulation, was outside the scope of this report.
4.6. Fill and finish requirements
Following the production of the mRNA vaccine drug substance, this active ingredient is filled into plastic bags and shipped to the fill and finish facilities, where it is filled into vials using aseptic filling lines operating under cGMP guidelines. The cost of filling into vials depends mostly on the filling technology and vial or container size. The cost per dose tends to decrease as the vial size increases. The NIH-Moderna mRNA-1273 is filled into 10-dose vials, whereas the BioNTech-Pfizer BNT162b2 vaccine is filled into 5-dose vials, which were eventually approved as 6-dose vials. While the vial size for CureVac’s CVnCoV is not known, given its thermostability,[66] larger multidose vials (e.g. 10-dose vials or higher) would be more cost-effective. The fill and finish production cost (including the cost of the vial) was estimated at $0.27 per dose and $0.37 per dose for filling into 10-dose vials and 5-dose vials, respectively. The cost estimates were obtained using the SuperPro Designer bioprocess modelling tool. Filling eight billion mRNA-1273 vaccine doses into 10-dose vials is estimated to cost $2.16 billion. Filling eight billion BNT162b2 vaccine doses into 5-dose (or 6-dose) vials is estimated to cost $3.04 billion, as shown below in Table 8.
It was estimated by the authors that approximately 15 vial filling lines are required to fill eight billion vaccine doses in 10-dose vials in six months. To fill eight billion vaccine doses in 5-dose vials in 6 months, approximately 25 vial filling lines are required. For these estimates a filling rate of 400 vials per minute and 5% losses in the fill and finish were assumed at 60% overall equipment effectiveness (OEE). The amount of empty glass vials required for 8 billion vaccine doses is 842.1 million vials and 1.684 billion vials for the 10-dose and 5-dose vials, respectively, after accounting for the 5% losses in the fill and finish.
Table 8: Key fill and finish resource requirements for producing 8 billion mRNA vaccine doses.
| Name of fill and finish resource | Fill and finish requirements for producing mRNA-1273 vaccines | Fill and finish requirements for producing BNT162b2 vaccines | Fill and finish requirements for producing CVnCoV vaccines |
| Doses per vial | 10 | 6 | 10** |
| Total number of filling lines for producing 8 billion doses* | 15 | 25 | 15 |
| Number of empty glass vials for producing 8 billion doses, with 5% losses | 842.1 million | 1.684 billion | 842.1 million |
| Total operating fill and finish costs for producing 8 billion doses*** | 2.16 billion | 3.04 billion | 2.16 billion |
* The values of the production requirements were calculated based on producing 8 billion vaccine doses within 6 months. ** The vial size for CureVac’s CVnCoV is not available publicly and it was assumed at 10 doses per vial, due to the higher thermostability of this vaccine [67]. *** Includes the cost of the empty glass vials, however, the cost of the drug substance is not included in this estimate. This includes the annualized capital cost for the fill and finish facilities.
5. Conclusion
In this report, we show one path forward on how to establish distributed mRNA vaccine production capacity for global pandemic response. This can be achieved rapidly by repurposing existing facilities, installing single-use production equipment, validating the process and starting-up production – all possible within six months. Following this, in the remaining six months the production of eight billion doses of vaccines can be achieved if sufficient resources are mobilized. The number of facilities, the amount of materials, consumables and labor required to produce eight billion vaccine doses depends on the three mRNA vaccines presented in this study. The mRNA-1273 vaccine is the most resource-intensive to produce, followed by the BNT162b2 vaccine and CVnCoV vaccine respectively.
In order to produce eight billion doses of the NIH-Moderna mRNA-1273 vaccine within six months, 55 production lines at the 30L bioreactor working volume scale are required. These 55 production lines can be placed in 14 geographically distributed manufacturing plants, with three or four production lines per facility. Producing eight billion doses of the BioNTech/Pfizer BNT162b2 vaccine within six months, requires 17 production lines, which can be placed in a total of five facilities, with three or four production lines per facility. The eight billion CureVac CVnCoV vaccine production target can be met in six months with seven production lines, housed in two facilities with three or four production lines per facility.
Appendix
Supplementary methods: Techno-economic modeling
Drug substance production (aka. primary manufacturing) modelling as well as drug product manufacturing (aka. fill and finish, secondary manufacturing) modelling has been carried out using SuperPro Designer Version 11, Build 2 from Intelligen, Inc. The input parameters and assumptions for drug substance and drug product techno-economic modelling in SuperPro Designer are listed in Table S1 below. Most of these parameters listed in Table S1 were kept at the default values from SuperPro Designer, as these default values are representative for biopharmaceutical production process and cost modelling. The Building Cost within the Direct Costs used for CapEx calculations was changed from the default value to 250% of the total equipment purchase cost (TEPC), as this value is more representative of GMP production processes which have higher facility costs. Updating this Building Cost to 250% of the TEPC was recommended by Demetri Petrides from Intelligen, Inc, who developed SuperPro Designer. The working capital cost period was decreased from the default value to 10 days because RNA vaccine production is faster compared to conventional cell-base biopharmaceutical production for which the default working capital cost period value was representative. The laboratory quality control (QC) and quality assurance (QA) costs were increased to 50% of the total labor costs (TLC) because this is a new technology and quality testing is likely to be more expensive compared to more established technologies. The impact of the QC/QA on the cost per dose was set at 50% of TLC the impact on the cost per dose was minimal. All production processes were modelled to operate 345 days per year. The time between consecutive batches (aka. cycle slack time) was set to 3 hours for production process models at the 30L scale. The number of campaigns per year was set to 1 in all the drug substance and drug product manufacturing models. The labor cost for drug substance production processes (operated in batch mode) was calculated using the detailed labor estimate, in function of the basic labor rate, benefits, operating supplies, supervision cost and administration cost.
Table S1: Input parameters and assumptions used for techno-economic modelling in SuperPro Designer.
| Parameter use | Parameter class | Parameter name | Value | Unit |
| CapEx calculation | Direct Cost
(DC) |
Piping Cost | 35 | % of TEPC |
| Instrumentation Cost | 40 | % of TEPC | ||
| Insulation Cost | 03 | % of TEPC | ||
| Electrical Facilities Cost | 10 | % of TEPC | ||
| Buildings Cost | 250 | % of TEPC | ||
| Yard Improvement Cost | 15 | % of TEPC | ||
| Auxiliary Facilities Cost | 40 | % of TEPC | ||
| Unlisted Equipment Purchase Cost (UEPC) | 30 | % of TEPC | ||
| Unlisted Equipment Installation Cost | 50 | % of UEPC | ||
| Indirect Cost (IC) | Engineering Cost | 25 | % of DC | |
| Construction Cost | 35 | % of DC | ||
| Other Cost (OC) | Contractor’s Fee | 5 | % of (IC + DC) | |
| Contingency | 10 | % of (IC + DC) | ||
| Miscellaneous | Working Capital – to cover expenses for | 10 mRNA & saRNA | days | |
| Start-up and Validation Costs | 30 | % of DFC | ||
| Up front R&D | 0 | US$ | ||
| Up front royalties | 0 | US$ | ||
| OpEx calculation | Facility dependent | Maintenance: equipment specific multipliers | ||
| Depreciation: contribution from each equipment’s undepreciated purchase cost | ||||
| Insurance | 1 | % of DFC | ||
| Local taxes | 2 | % of DFC | ||
| Factory expenses | 5 | % of DFC | ||
| Labor | Basic operator labor rate (BOLR) | 35 | USD × hour-1 | |
| Benefits factor | 40 | % of BOLR | ||
| Operating supplies factor | 10 | % of BOLR | ||
| Supervision factor | 20 | % of BOLR | ||
| Administration factor | 60 | % of BOLR | ||
| Lumped operator labor rate | 80.5 | USD × hour-1 | ||
| Adjusted basic operator labor rate* | 80.5 | USD × hour-1 | ||
| Direct labor time utilization – batch | 60 | % | ||
| Direct labor time utilization – continuous | 70 | % | ||
| Lab, QC, QA | Laboratory, quality control, quality assurance | 50 | % TLC | |
| Utilities | Standard electricity | 0.1 | US$× (kW×h)-1 | |
| Chilled water | 0.4 | US$ × tonne-1 | ||
| Cooled water | 0.1 | US$ × tonne-1 | ||
| Steam | 12 | US$ × tonne-1 | ||
| Water for injection | 0.12 | US$ × kg-1 | ||
| Miscellaneous | Fixed R&D | 0 | US$ × year-1 | |
| Variable R&D | 0 | US$ × g MP-1 | ||
| On-going process validation | 0 | US$ × year-1 | ||
| Other fixed | 0 | US$ × year-1 | ||
| Other variable | 0 | US$ × g MP-1 | ||
| Overall economic evaluation | Time valuation | Construction period | 20 | months |
| Start-up period | 4 | months | ||
| Project lifetime | 20 | years | ||
| Inflation | 4 | % | ||
| NPV interest – Low | 7 | % | ||
| NPV interest – Medium | 9 | % | ||
| NPV interest – High | 11 | % | ||
| Financing | Loan interest for DFC | 9 | % | |
| Loan interest for working capital | 12 | % | ||
| Loan interest for up front R&D | 12 | % | ||
| Loan interest for up front royalties | 12 | % | ||
| Loan period for DFC | 10 | years | ||
| Loan period for working capital | 6 | years | ||
| Loan period for up front R&D | 6 | years | ||
| Loan period for up front royalties | 6 | years | ||
| DFC outlay for 1st year | 30 | % of DFC | ||
| DFC outlay for 2nd year | 40 | % of DFC | ||
| DFC outlay for 3rd year | 30 | % of DFC | ||
| DFC outlay for 4th year | 0 | % of DFC | ||
| DFC outlay for 5th year | 0 | % of DFC | ||
| Straight line depreciation period | 10 | years | ||
| Salvage value | 5 | % of DFC | ||
| Production level | Operating capacity for each year | 100 | % | |
| Product failure rate | 5 | % | ||
| Disposal cost | 0 | US$ × g MP-1 | ||
| Miscellaneous | Income tax | 40 | % | |
| Fixed advertising and selling expenses | 0 | US$ × year-1 | ||
| Variable advertising and selling expenses | 0 | US$ × g MP-1 | ||
| Variable running royalty expenses | 0 | US$ × g MP-1 | ||
Abbreviations used in Table S1: CapEx – capital expenditure; OpEx – operating expense; TEPC – total equipment purchase cost; UEPC – unlisted equipment purchase cost; DFC – direct fixed capital; DC – direct cost; IC – indirect cost; OC – other cost; TLC – total labor costs; BOLR – basic operator labor rate; g MP – gram of main product. *calculated based on benefits, operating supplies, supervision cost and administration cost.
Supplementary results
Table S2. The estimated costs and doses produced per year per drug substance production line for the NIH-Moderna (mRNA-1273), BioNTech-Pfizer (BNT162b2), and CureVac (CVnCoV) vaccines.
| Name of indication | Amount per production line for mRNA-1273 vaccine drug substance production * | Amount per production line for BNT162b2 vaccine drug substance production * | Amount per production line for CVnCoV vaccine drug substance production * |
| Capital cost, CapEx [USD] | 57,947,000 | 57,947,000 | 57,947,000 |
| Operating cost, OpEx [USD/year] | 635,690,000 | 635,690,000 | 635,690,000 |
| Doses per year | 291,620,000 | 972,066,667 | 2,430,166,667 |
* Based on production at the 30L bioreactor working volume scale, producing 29162 grams of mRNA per year.
Table S3: Estimated amounts of raw materials used in the mRNA-LNP vaccine drug substance production process based on the manufacturing process illustrated in Figure 2. **
| Name of pure component | Material amounts per year per production line*
[g / year] |
Material amount for producing 8 billion mRNA-1273 doses
[g / 8 billion doses] |
Material amount for producing 8 billion BNT162b2 doses
[g / 8 billion doses] |
Material amount for producing 8 billion CVnCoV doses
[g / 8 billion doses] |
| Acetic acid | 9,926,646 | 286,649,349 | 85,994,805 | 34,397,922 |
| Adenosine-5′-triphosphate (ATP) | 22,519 | 650,276 | 195,083 | 78,033 |
| Calcium chloride (CaCl2) | 5,977 | 172,596 | 51,779 | 20,712 |
| Cholesterol | 187,675 | 5,419,445 | 1,625,834 | 650,333 |
| Citric acid | 1,357,085 | 39,188,214 | 11,756,464 | 4,702,586 |
| CleanCap AG | 50,972 | 1,471,906 | 441,572 | 176,629 |
| cytidine-5′-triphosphate (CTP) | 21,452 | 619,464 | 185,839 | 74,336 |
| Deoxyribonuclease I (DNase I)*** | 28 | 809 | 243 | 97 |
| Disodium phosphate (Na2HPO4) | 17,130,535 | 494,674,304 | 148,402,291 | 59,360,916 |
| Dithiothreitol (DTT) | 18,193 | 525,355 | 157,606 | 63,043 |
| Ethyl alcohol (ethanol) | 93,049,356 | 2,686,963,682 | 806,089,105 | 322,435,642 |
| Guanosine-5′-triphosphate (GTP) | 23,229 | 670,778 | 201,233 | 80,493 |
| Ionizable lipid | 434,380 | 12,543,486 | 3,763,046 | 1,505,218 |
| Linear template DNA | 555 | 16,027 | 4,808 | 1,923 |
| Magnesium chloride (MgCl2) | 30,890 | 892,003 | 267,601 | 107,040 |
| Monopotassium phosphate (KH2PO4) | 31,146 | 899,395 | 269,819 | 107,927 |
| Phospholipid | 94,139 | 2,718,429 | 815,529 | 326,211 |
| Polyethylene glycol (PEG) lipid | 53,576 | 1,547,101 | 464,130 | 185,652 |
| Potassium chloride (KCl) | 30,119,051 | 869,740,530 | 260,922,159 | 104,368,864 |
| Pyrophosphatase*** | 888 | 25,643 | 7,693 | 3,077 |
| RNase enzyme inhibitor | 139 | 4,014 | 1,204 | 482 |
| Sodium acetate | 46,324,346 | 1,337,696,902 | 401,309,071 | 160,523,628 |
| Sodium chloride (NaCl) | 124,585,712 | 3,597,631,384 | 1,079,289,415 | 431,715,766 |
| Sodium citrate | 7,291,590 | 210,557,476 | 63,167,243 | 25,266,897 |
| Sodium hydroxide (NaOH) | 8,795,066 | 253,972,987 | 76,191,896 | 30,476,758 |
| Spermidine | 3,225 | 93,128 | 27,938 | 11,175 |
| Sucrose | 761,772 | 21,997,505 | 6,599,252 | 2,639,701 |
| T7 RNA polymerase*** | 185 | 5,342 | 1,603 | 641 |
| Tris hydrochloride (Tris HCl) | 272,325 | 7,863,863 | 2,359,159 | 943,664 |
| Water for injection (WFI), RNAse free | 17,811,917,463 | 514,350,419,836 | 154,305,125,951 | 61,722,050,380 |
| 1-methylpseudouridine-5′-triphosphate (mod-UTP) | 21,496 | 620,735 | 186,220 | 0 |
* Production line at 30L bioreactor working volume scale, producing 29162 grams of mRNA per year. ** For calculating the material requirements for producing 8 billion doses and additional 5% losses were assumed to occur in the fill and finish process. This is the list of all materials used in the production process, including materials used for cleaning. *** The amount of enzymes required depends on the specific activity of the enzymes and this can vary between different suppliers.
Table S4: Estimated cost of raw materials used in the mRNA-LNP vaccine drug substance production process based on the manufacturing process illustrated in Figure 2. **
| Name of solution or material | Material amounts per year per production line*
[kg/year] |
Annual material cost per production line*
[USD/year] |
Material costs for producing 8 billion mRNA-1273 doses
[USD / 8 billion doses] |
Material costs for producing 8 billion BNT162b2 doses
[USD / 8 billion doses] |
Material costs for producing 8 billion CVnCoV doses
[USD / 8 billion doses] |
||
| 0.1 M CaCl2 solution | 23 | 133 | 3,841 | 1,152 | 461 | ||
| 0.1 M Spermidine solution | 221 | 27,112 | 782,907 | 234,872 | 93,949 | ||
| 1 M DTT solution | 126 | 109,170 | 3,152,476 | 945,743 | 378,297 | ||
| 1 M MgCl2 solution | 66 | 204 | 5,891 | 1,767 | 707 | ||
| 1 M NaOH solution | 13,616 | 1,571 | 45,365 | 13,610 | 5,444 | ||
| 1 M Tris HCl solution | 961 | 98,887 | 2,855,536 | 856,661 | 342,664 | ||
| 1 mg/ml DNA template solution | 552 | 61,050,086 | 1,762,928,524 | 528,878,557 | 211,551,423 | ||
| 100 mM ATP solution | 442 | 5,549,985 | 160,265,571 | 48,079,671 | 19,231,868 | ||
| 100 mM CleanCap AG solution | 449 | 203,887,446 | 5,887,608,320 | 1,766,282,496 | 706,512,998 | ||
| 100 mM CTP solution | 442 | 5,538,693 | 159,939,494 | 47,981,848 | 19,192,739 | ||
| 100 mM GTP solution | 442 | 5,549,984 | 160,265,542 | 48,079,663 | 19,231,865 | ||
| 100 mM mod-UTP solution | 442 | 101,528,497 | 2,931,813,779 | 879,544,134 | 351,817,654 | ||
| 250 mM KCl solution | 1,621,076 | 551,973 | 15,939,190 | 4,781,757 | 1,912,703 | ||
| Cholesterol | 188 | 4,691,877 | 135,486,194 | 40,645,858 | 16,258,343 | ||
| DNase I | 11 | 14,764 | 426,336 | 127,901 | 51,160 | ||
| Ethyl Alcohol | 93,049 | 744,395 | 21,495,714 | 6,448,714 | 2,579,486 | ||
| Ionizable lipid | 434 | 21,719,000 | 627,174,294 | 188,152,288 | 75,260,915 | ||
| Sodium Citrate buffer | 405,377 | 200,225 | 5,781,849 | 1,734,555 | 693,822 | ||
| PBS solution | 15,573,214 | 1,877,842 | 54,225,988 | 16,267,796 | 6,507,119 | ||
| PEG lipid | 54 | 1,071,520 | 30,942,023 | 9,282,607 | 3,713,043 | ||
| Phospholipid | 94 | 1,412,086 | 40,776,465 | 12,232,939 | 4,893,176 | ||
| Pyrophosphatase solution | 468 | 14,714,660 | 424,911,667 | 127,473,500 | 50,989,400 | ||
| RNase enzyme inhibitor solution | 276 | 118,137 | 3,411,413 | 1,023,424 | 409,370 | ||
| Sodium acetate | 330,888 | 766,536 | 22,135,074 | 6,640,522 | 2,656,209 | ||
| Sucrose | 762 | 3,809 | 109,992 | 32,997 | 13,199 | ||
| T7 RNA polymerase solution | 184 | 25,216,706 | 728,176,702 | 218,453,011 | 87,381,204 | ||
| Tris-HCl 1x buffer | 102,680 | 162,239 | 4,684,936 | 1,405,481 | 562,192 | ||
| Water for injection (WFI), RNAse free | 6,178 | 741 | 21,398 | 6,419 | 2,568 | ||
| TOTAL | 18,152,715 | 456,608,278 | 13,185,366,482 | 3,955,609,944 | 1,582,243,978 | ||
* Production line at 30L bioreactor working volume scale, producing 29162 grams of mRNA per year. ** For calculating the material requirements for producing 8 billion doses and additional 5% losses were assumed to occur in the fill and finish process. This is the list of materials used in the production process which come in direct contact with the mRNA product, excluding auxiliary materials (e.g. materials used for cleaning). In addition to these costs there might also be additional licensing costs for using the lipids in the LNP formulation step, especially for the ionizable lipid.
Acknowledgments
This report was written by Dr. Zoltán Kis and Zain Rizvi. It was edited by Peter Maybarduk, Rhoda Feng and Josephine Fonger at Public Citizen.
Dr. Kis is a Research Associate, Centre for Process Systems Engineering, Department of Chemical Engineering, Imperial College London. His work on this report employs tools and datasets that he developed previously in work funded by the Department of Health and Social Care using UK Aid funding, managed by the Engineering and Physical Sciences Research Council (EPSRC, grant number: EP/R013764/1).
Mr. Rizvi is a law and policy researcher in Public Citizen’s Access to Medicines Program.
Public Citizen commissioned this analysis from Dr. Kis as part of a consultancy project. Dr. Kis gratefully acknowledges insightful discussions with Prof. Nilay Shah (Imperial College London, UK), Dr. Cleo Kontoravdi (Imperial College London, UK) and John Liddell (Centre for Process Innovation, UK).
References
[1] BioNTech, ‘BioNTech to Acquire GMP Manufacturing Site to Expand COVID-19 Vaccine Production Capacity in First Half 2021’, BioNTech SE, 2020 <https://investors.biontech.de/news-releases/news-release-details/biontech-acquire-gmp-manufacturing-site-expand-covid-19-vaccine> [accessed 20 November 2020].
[2] Erika Solomon, ‘“Where the Magic Happens” — inside BioNTech’s Innovative Vaccine Plant’, Financial Times, 2021 <https://www.ft.com/content/cf5d6113-3698-4cc7-9d5b-8f0f29fd6a35> [accessed 14 May 2021].
[3] BioNTech, ‘BioNTech to Acquire GMP Manufacturing Site to Expand COVID-19 Vaccine Production Capacity in First Half 2021’; Pharmaceutical Technology, ‘BioNTech’s COVID-19 Vaccine Manufacturing Facility, Marburg’, Verdict Media Limited, 2021; James; Krellenstein and Christian Urrutia, Hit Hard, Hit Fast, Hit Globally – A Model for Global Vaccine Access (New York, NY, USA, 2021); BioNTech, ‘BioNTech Provides Update on Vaccine Production Status at Marburg Manufacturing Site’, BioNTech SE, 2021 [accessed 14 May 2021].
[4] This report builds on previously published work by one of the authors. Zoltán Kis, Cleo Kontoravdi, Robin Shattock, and others, ‘Resources, Production Scales and Time Required for Producing RNA Vaccines for the Global Pandemic Demand’, Vaccines, 2021, 1–14 <https://doi.org/10.3390/vaccines9010003>; Zoltán Kis, Cleo Kontoravdi, Antu K Dey, and others, ‘Rapid Development and Deployment of High‐volume Vaccines for Pandemic Response’, Journal of Advanced Manufacturing and Processing, 2.3 (2020), e10060 <https://doi.org/10.1002/amp2.10060>. The biomanufacturing models presented therein have been updated to include the latest developments in this rapidly evolving field. Krellenstein and Urrutia.
[5] The World Bank estimates 5 billion people live in low-and-middle income countries, excluding China (6.4 bn – 1.4 billion). Assuming herd immunity is 80 percent, 4 billion people need vaccines. With a two-dose vaccine, this means 8 billion doses are required.
[6] The CureVac candidate is still in clinical trials and has not received emergency use authorization.
[7] If Moderna received regulatory authorization for a lower 50 µg mRNA dose, it could significantly reduce costs.
[8] Pharmaceutical Technology; Krellenstein and Urrutia.
[9] World Health Organization, 6th Access to COVID-19 Tools (ACT) Accelerator Facilitation Council meeting <https://www.who.int/news-room/events/detail/2021/05/12/default-calendar/6th-access-to-covid-19-tools-(act)-accelerator-facilitation-council-meeting> (at 45:51)
[10] U.S. Food and Drug Administration (FDA), ‘Current Good Manufacturing Practice (CGMP) Regulations, 21 CFR Part 210, 211, 600, <https://www.fda.gov/drugs/pharmaceutical-quality-resources/current-good-manufacturing-practice-cgmp-regulations> [accessed 27 April 2021]; World Health Organization, Annex 2 – WHO Good Manufacturing Practices for Pharmaceutical (Geneva, Switzerland, 2016) <https://www.who.int/publications/m/item/annex-2-WHO-gmp-for-biological-products>; ICH Expert Working Group, ICH Harmonised Tripartite Guideline on Good Manufacturing Practice Guide for Active Pharmaceutical Ingredients Q7 (Geneva, Switzerland, 2000) <https://database.ich.org/sites/default/files/Q7_Guideline.pdf>.
[11] Zoltán Kis, Cleo Kontoravdi, Robin Shattock, and others, ‘Resources, Production Scales and Time Required for Producing RNA Vaccines for the Global Pandemic Demand’, Vaccines, 2021, 1–14 <https://doi.org/10.3390/vaccines9010003>; Zoltán Kis, Cleo Kontoravdi, Antu K Dey, and others, ‘Rapid Development and Deployment of High‐volume Vaccines for Pandemic Response’, Journal of Advanced Manufacturing and Processing, 2.3 (2020), e10060 <https://doi.org/10.1002/amp2.10060>.
[12] E P Wen, R J Ellis, and N S Pujar, Vaccine Development and Manufacturing, Wiley Series in Biotechnology and Bioengineering (Hoboken, New Jersey, United States: John Wiley & Sons, Inc., 2015) <https://doi.org/10.1002/9781118870914>; Kis, Shattock, Shah, and others.
[13] Wen, Ellis, and Pujar; Kis, Shattock, Shah, and others; Joanna Sugden, ‘Oxford-AstraZeneca’s Covid-19 Vaccine Helps U.K. Lead Race to Reach Nursing Homes’, The Wall Street Journal, 2021 <https://www.wsj.com/articles/oxford-astrazenecas-covid-19-vaccine-helps-u-k-lead-race-to-reach-nursing-homes-11611138601> [accessed 28 April 2021].
[14] D L Nelson and M M Cox, Lehninger Principles of Biochemistry, 6th edn (Macmillan Learning, 2012) <https://books.google.co.uk/books?id=n9e1NAEACAAJ>; Wen, Ellis, and Pujar; Deutsche Welle, ‘AstraZeneca COVID Vaccine’s Complex EU Supply Chain’, Deutsche Welle – Business, 2021 <https://www.dw.com/en/astrazeneca-covid-vaccine-oxford/a-56427963> [accessed 12 May 2021]; Jenny; Strasburg and Laurence Norman, ‘Behind AstraZeneca’s Covid-19 Vaccine Stumble’, The Wall Street Journal, 2021 <https://www.wsj.com/articles/behind-astrazenecas-covid-19-vaccine-stumble-11611871968> [accessed 13 May 2021].
[15] Kis, Kontoravdi, Dey, and others; Kis, Kontoravdi, Shattock, and others.
[16] Alice Park and Aryn Baker, ‘Exclusive: Inside the Facilities Making the World’s Most Prevalent COVID-19 Vaccine’, Time, 2021 <https://time.com/5955247/inside-biontech-vaccine-facility/> [accessed 27 April 2021].
[17] Sugden; Wen, Ellis, and Pujar.
[18] Kis, Kontoravdi, Dey, and others; Kis, Shattock, Shah, and others.
[19] Wen, Ellis, and Pujar; Park and Baker.
[20] Park and Baker.
[21] World Health Organization, Environmental Monitoring of Clean Rooms in
Vaccine Manufacturing Facilities (Geneva, Switzerland, 2016) <https://www.who.int/publications/m/item/annex-2-WHO-gmp-for-biological-products> (noting differences in clean room requirements for open and closed systems)
[22] Norbert Pardi and others, ‘MRNA Vaccines — a New Era in Vaccinology’, Nature Reviews. Drug Discovery, 17.4 (2018), 261–79 <https://doi.org/10.1038/nrd.2017.243>.
[23] Hopkins Jared, ‘Pfizer Lifts Covid-19 Vaccine Production Targets for 2021, 2022,’ The Wall Street Journal <https://www.wsj.com/articles/pfizer-lifts-covid-19-vaccine-production-targets-for-2021-2022-11620425904>.
[24] Ugur Sahin and others, ‘COVID-19 Vaccine BNT162b1 Elicits Human Antibody and TH1 T Cell Responses’, Nature, 586.7830 (2020), 594–99 <https://doi.org/10.1038/s41586-020-2814-7>; Katalin Karikó and others, ‘Incorporation of Pseudouridine into MRNA Yields Superior Nonimmunogenic Vector with Increased Translational Capacity and Biological Stability.’, Molecular Therapy : The Journal of the American Society of Gene Therapy, 16.11 (2008), 1833–40 <https://doi.org/10.1038/mt.2008.200>.
[25] Bancel and others; Scorza Francesco Berlanda and others; Heartlein and others; Funkner and others; Kis, Shattock, Shah, and others; Kis, Kontoravdi, Dey, and others; Centre for Process Innovation Limited; Kis, Kontoravdi, Shattock, and others; Gagnon, Purification of Nucleic Acids – A Handbook for Purification of DNA Plasmids and MRNA for Gene Therapy and Vaccines; Gagnon, ‘A New Runway for Purification of Messenger RNA’.
[26] Bancel and others; Scorza Francesco Berlanda and others; Heartlein and others; Funkner and others; Kis, Shattock, Shah, and others; Kis, Kontoravdi, Dey, and others; Centre for Process Innovation Limited; Kis, Kontoravdi, Shattock, and others; Gagnon, Purification of Nucleic Acids – A Handbook for Purification of DNA Plasmids and MRNA for Gene Therapy and Vaccines; Gagnon, ‘A New Runway for Purification of Messenger RNA’.
[27] Demetri Petrides, SuperPro Designer User Guide – A Comprehensive Simulation Tool for the Design, Retrofit & Evaluation of Specialty Chemical, Biochemical, Pharmaceutical, Consumer Product, Food, Agricultural, Mineral Processing, Packaging AND Water Purification, Wastewater (Scotch Plains, NJ, USA, 2013) <http://www.intelligen.com/downloads/SuperPro_ManualForPrinting_v10.pdf (accessed on 22.Mar.2020)>; Demetri Petrides, ‘Bioprocess Design and Economics’, in Bioseparations Science and Engineering, 2nd Editio (Oxford, UK: Oxford University Press, 2015), pp. 1–83 <http://www.intelligen.com/downloads/BioProcessDesignAndEconomics_March_2015.pdf (accessed on 12.May.2020)>; Kis, Kontoravdi, Shattock, and others.
[28] Cytiva, CaptoTM Core 700 Chromatography (Uppsala, Sweden, 2020) <https://cdn.cytivalifesciences.com/dmm3bwsv3/AssetStream.aspx?mediaformatid=10061&destinationid=10016&assetid=16124>; Intellectual Property Notice, The Biopharma, and G E Healthcare, CaptoTM Core 700 in Vaccine Processing (Uppsala, Sweden, 2020) <https://cdn.cytivalifesciences.com/dmm3bwsv3/AssetStream.aspx?mediaformatid=10061&destinationid=10016&assetid=17547>.
[29] Michael D Buschmann and others, ‘Nanomaterial Delivery Systems for MRNA Vaccines’, Vaccines, 2021 <https://doi.org/10.3390/vaccines9010065>.
[30] Buschmann and others; Amanda Sealy, ‘Manufacturing Moonshot: How Pfizer Makes Its Millions of Covid-19 Vaccine Doses’, CNN, 2021 <https://edition.cnn.com/2021/03/31/health/pfizer-vaccine-manufacturing/index.html> [accessed 27 April 2021].
[31] Park and Baker.
[32] Buschmann and others.
[33] Park and Baker.
[34] BioNTech, ‘Next Generation Immunotherapy’ (Mainz, Germany: BioNTech SE, 2021), pp. 1–59 <https://investors.biontech.de/static-files/53947c63-dd5c-44d3-8543-87c1ecad96a1>.
[35] BioNTech, ‘BioNTech to Acquire GMP Manufacturing Site to Expand COVID-19 Vaccine Production Capacity in First Half 2021’; Pharmaceutical Technology; Krellenstein and Urrutia; BioNTech, ‘BioNTech Provides Update on Vaccine Production Status at Marburg Manufacturing Site’.
[36] Sealy.
[37] FDA, Emergency Use Authorization for Vaccines to Prevent COVID-19; U.S. Food and Drug Administration (FDA), <https://www.fda.gov/regulatory-information/search-fda-guidance-documents/emergency-use-authorization-vaccines-prevent-covid-19>.
[38] FDA, Emergency Use Authorization for Vaccines to Prevent COVID-19; U.S. Food and Drug Administration (FDA), <https://www.fda.gov/regulatory-information/search-fda-guidance-documents/emergency-use-authorization-vaccines-prevent-covid-19>.
[39] Pharmaceutical Technology; Krellenstein and Urrutia.
[40] Sealy; Krellenstein and Urrutia.
[41] The scale of the production processes was expressed based on the working volume of the production bioreactor and the scale in this study was fixed at the 30L bioreactor working volume scale. The production batches were scheduled for maximising equipment utilisation, thus maximising the number of production batches per year. However, the production process was not de-bottlenecked.
[42] Petrides, ‘Bioprocess Design and Economics’; Demetri Petrides and others, ‘Biopharmaceutical Process Optimization with Simulation and Scheduling Tools’, Bioengineering (Basel, Switzerland: MDPI, 2014), 154–87 <https://doi.org/10.3390/bioengineering1040154>; Petrides, SuperPro Designer User Guide – A Comprehensive Simulation Tool for the Design, Retrofit & Evaluation of Specialty Chemical, Biochemical, Pharmaceutical, Consumer Product, Food, Agricultural, Mineral Processing, Packaging AND Water Purification, Wastewater.
[43] Kis, Kontoravdi, Shattock, and others; Kis, Kontoravdi, Dey, and others.
[44] European Medicines Agency, Assessment Report – COVID-19 Vaccine Moderna (Amsterdam, The Netherlands, 2021), EMA/15689/ <https://www.ema.europa.eu/en/documents/assessment-report/covid-19-vaccine-moderna-epar-public-assessment-report_en.pdf>; U.S. Food and Drug Administration (FDA), ‘Fact Sheet for Healthcare Providers Administering – Vaccine (Vaccination Providers) – Emergency Use Authorization (EUA) of the Moderna Covid-19 Vaccine To Prevent Coronavirus Disease 2019 (Covid-19)’, U.S. FDA, 2020, pp. 1–22 <https://www.fda.gov/media/144637/download> [accessed 25 January 2021].
[45] U.S. Food and Drug Administration (FDA), Fact Sheet for Healthcare Providers Administering Vaccine (Vaccination Providers) – Emergency Use Authorization (EUA) of the Pfizer-BioNTech Covid-19 Vaccine to Prevent Coronavirus Disease 2019 (Covid-19), U.S. FDA, 2021, viii <https://www.fda.gov/media/144413/download>; Pfizer Manufacturing Belgium NV, ‘Pfizer-BioNTech Covid-19 Vaccine Fact Sheet for Healthcare Providers Administering Vaccine (Vaccination Providers) – Emergency Use Authorization (EUA) of the Pfizer-BioNTech Covid-19 Vaccine to Prevent Coronavirus Disease 2019 (Covid-19)’, 2021 <http://labeling.pfizer.com/ShowLabeling.aspx?id=14471> [accessed 20 April 2021].
[46] Buschmann and others; CureVac, Clinical Trial Protocol, COVID-19: A Phase 2b/3, Randomized, Observer-Blinded, Placebo-Controlled, Multicenter Clinical Study Evaluating the Efficacy and Safety of Investigational SARS-CoV-2 MRNA Vaccine CVnCoV in Adults 18 Years of Age and Older (Frankfurt, Germany, 2020) <https://www.curevac.com/wp-content/uploads/2020/12/20201214-CureVac-HERALD-Clinical-Trial-Protocol-of-Phase-2b_3_CVnCoV.pdf>.
[47] BioNTech, ‘BioNTech to Acquire GMP Manufacturing Site to Expand COVID-19 Vaccine Production Capacity in First Half 2021’; Pharmaceutical Technology; Krellenstein and Urrutia; BioNTech, ‘BioNTech Provides Update on Vaccine Production Status at Marburg Manufacturing Site’.
[48] Sealy.
[49] Sealy.
[50] Jeffery Odum, ‘Closed Systems in Bioprocessing’, Encyclopedia of Industrial Biotechnology, Major Reference Works, 2010, pp. 1–6 <https://doi.org/doi:10.1002/9780470054581.eib235>.
[51] Pharmaceutical Technology; Krellenstein and Urrutia.
[52] If 5’ capping of the mRNA is carried out enzymatically, the amounts of capping enzymes should be considered instead of the 5’ cap analogues (e.g. CleanCap AG).
[53] European Medicines Agency, EMA/15689/; U.S. Food and Drug Administration (FDA), ‘Fact Sheet for Healthcare Providers Administering – Vaccine (Vaccination Providers) – Emergency Use Authorization (EUA) of the Moderna Covid-19 Vaccine To Prevent Coronavirus Disease 2019 (Covid-19)’; U.S. Food and Drug Administration (FDA), viii; Pfizer Manufacturing Belgium NV; Buschmann and others; CureVac, Clinical Trial Protocol, COVID-19: A Phase 2b/3, Randomized, Observer-Blinded, Placebo-Controlled, Multicenter Clinical Study Evaluating the Efficacy and Safety of Investigational SARS-CoV-2 MRNA Vaccine CVnCoV in Adults 18 Years of Age and Older.
[54] Buschmann and others.
[55] European Medicines Agency, EMA/15689/; U.S. Food and Drug Administration (FDA), ‘Fact Sheet for Healthcare Providers Administering – Vaccine (Vaccination Providers) – Emergency Use Authorization (EUA) of the Moderna Covid-19 Vaccine To Prevent Coronavirus Disease 2019 (Covid-19)’; U.S. Food and Drug Administration (FDA), viii; Pfizer Manufacturing Belgium NV; Buschmann and others; CureVac, Clinical Trial Protocol, COVID-19: A Phase 2b/3, Randomized, Observer-Blinded, Placebo-Controlled, Multicenter Clinical Study Evaluating the Efficacy and Safety of Investigational SARS-CoV-2 MRNA Vaccine CVnCoV in Adults 18 Years of Age and Older.
[56] Buschmann and others.
[57] European Medicines Agency, EMA/15689/; U.S. Food and Drug Administration (FDA), ‘Fact Sheet for Healthcare Providers Administering – Vaccine (Vaccination Providers) – Emergency Use Authorization (EUA) of the Moderna Covid-19 Vaccine To Prevent Coronavirus Disease 2019 (Covid-19)’.
[58] U.S. Food and Drug Administration (FDA), viii; Pfizer Manufacturing Belgium NV.
[59] European Medicines Agency, EMA/15689/; U.S. Food and Drug Administration (FDA), ‘Fact Sheet for Healthcare Providers Administering – Vaccine (Vaccination Providers) – Emergency Use Authorization (EUA) of the Moderna Covid-19 Vaccine To Prevent Coronavirus Disease 2019 (Covid-19)’.
[60] U.S. Food and Drug Administration (FDA), viii; Pfizer Manufacturing Belgium NV.
[61] Buschmann and others; CureVac, Clinical Trial Protocol, COVID-19: A Phase 2b/3, Randomized, Observer-Blinded, Placebo-Controlled, Multicenter Clinical Study Evaluating the Efficacy and Safety of Investigational SARS-CoV-2 MRNA Vaccine CVnCoV in Adults 18 Years of Age and Older.
[62] Thermofisher Scientific, ‘Product Information Sheet – POROSTM Oligo ( DT ) 25 Affinity Resin’, Thermofisher Scientific, 2020, pp. 1–8 <https://www.thermofisher.com/order/catalog/product/A47382#/A47382> [accessed 12 May 2021].
[63] Edmond Girasek and others, ‘Headcount and FTE Data in the European Health Workforce Monitoring and Planning Process’, Human Resources for Health, 14.Suppl 1 (2016), 42 <https://doi.org/10.1186/s12960-016-0139-2>.
[64] John Miller, ‘Exclusive: Lonza Sets New Goal to Make Moderna COVID-19 Vaccine Ingredients’, Reuters, 2021 <https://www.reuters.com/article/us-health-coronavirus-vaccine-lonza-excl/exclusive-lonza-sets-new-goal-to-make-moderna-covid-19-vaccine-ingredients-idUSKBN2392C4> [accessed 26 April 2021]; John Miller, ‘Help Wanted: Lonza Seeks Workers to Lift Moderna Vaccine Output’, Reuters, 2021 <https://www.reuters.com/business/healthcare-pharmaceuticals/switzerlands-lonza-boost-production-moderna-covid-19-vaccine-2021-04-29/> [accessed 12 May 2021].
[65] Pharmaceutical Technology; Krellenstein and Urrutia.
[66] CureVac, ‘CureVac’s COVID-19 Vaccine Candidate, CVnCoV, Suitable for Standard Fridge Temperature Logistics’, CureVac AG, Tübingen, Germany, 2020 <https://www.curevac.com/en/2020/11/12/curevacs-covid-19-vaccine-candidate-cvncov-suitable-for-standard-fridge-temperature-logistics/> [accessed 14 March 2021].
[67] CureVac, ‘CureVac’s COVID-19 Vaccine Candidate, CVnCoV, Suitable for Standard Fridge Temperature Logistics’.